...on the rationalisation, design, optimisation and synthesis of novel phosphonium derived anti-cancer drugs
 |
Establishing Anti-Cancer Phosphonium Salt Structure-activity RelationshipsPart 2by Phil |
Delivery to the Mitochondria
2.1 The Nernst-Born model: Targeting cations to the mitochondria
Cations accumulate to mitochondria because being charged, they localise to regions of low electrochemical potential. [36] The force which drives this migration is governed by Coulombs law and the energy change [37] is governed by the Nernst equation (Equation 1).
Equation 1 | The Nernst equation
$$\Delta {\psi _m} = \frac{{ - \Delta G}}{{zF}}$$
…describes the linear variation of electrochemical potential with Gibbs free energy |
Equation 1 defines the electrochemical potential (Δψm) in terms of the Gibbs free energy. For typical mitochondrial activity, the cation is the proton and the energy change drives ATPsynthase [38] – however this is a universal force and all cations are attracted to the matrix.
Equation 2 | The Gibbs function
$$\Delta G = - RT\ln K = - RT\ln \frac{{{{[{X^ + }]}_{in}}}}{{{{[{X^ + }]}_{out}}}}$$
… the definition of Gibbs free energy in terms of equilibrium constant of a cation, X
From Gibbs equation we can calculate the cation equilibrium partition concentrations – the greater the energy change, the greater the drive to accumulate. This is embodied by Equation 3, a combination of Equation 1 & Equation 2.
Equation 3 | Equilibrium distribution related to Δψm
$$zF\Delta {\psi _m} = \Delta G = - RT\ln \frac{{{{[{X^ + }]}_{in}}}}{{{{[{X^ + }]}_{out}}}}$$
Accumulation is therefore a function of the charge and the membrane potential. For monovalent cations the Nernst equation in a form applicable to cell membranes can be simplified to give Equation 4. This is the Nernst equation in a form which determines accumulation as an electrochemical function.
Equation 4 | Simplified Nernst equation for monocations under physiological conditions
$$\Delta {\psi _m} = \frac{{61.5}}{z} \times \log \left( {\frac{{\left[ {cation} \right]_{in}^{}}}{{\left[ {cation} \right]_{out}^{}}}} \right)$$
2.2 Relative Cation Accumulation and its role in Cancer Selectivity
The above derivation maybe somewhat simplified taking into account only solvational effects but it does provide insight into the variables determining uptake and accumulation. The extent of accumulation can be calculated by solution of Equation 4 to yield Equation 5.
Equation 5 | Accumulation as a function of charge and Δψm
$${10^{{\raise0.5ex\hbox{$\scriptstyle {z
\times \Delta {\psi _m}}$}
\kern-0.1em/\kern-0.15em
\lower0.25ex\hbox{$\scriptstyle {61.5}$}}}} =
\frac{{{{[cation]}_{matrix}}}}{{{{[cation]}_{plasma}}}}$$
Graph 1 shows how accumulation varies across a range of Δψm (ie., physiological and mitochondrial membranes) for variously charged cations.
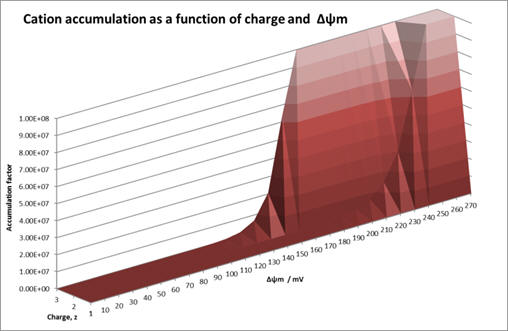
Graph 1 | Accumulation as a function of charge and membrane potential
The accumulation factor rises exponentially with charge: accumulation for +2 and +3 is exceedingly high even at low membrane potentials. Whilst this corresponds to a huge drug accumulation there is no selectivity. Agents must be able to discriminate between healthy and cancer mitochondria over the range of physiological potentials, ie., -200 and -270mV (see Graph 2) and monocations are most suitable.
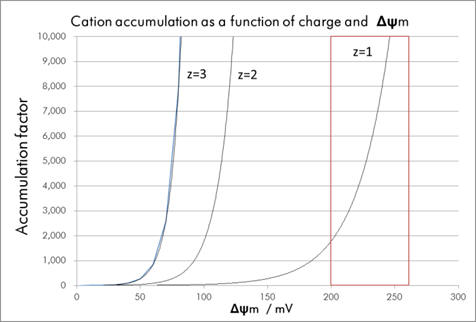
Graph 2 | Accumulation as a function of membrane potential for variously charged cations
Highly charged cations may be appealing by virtue of their
accumulation but for kinetics reasons, as will be outlined shortly,
highly charged cations suffer poor uptake.
Using Equation 5 the
equilibrium accumulation of cations into the different physiological
compartments (see Table 1 on page 8) can be calculated as a function
of their respective membrane potentials.
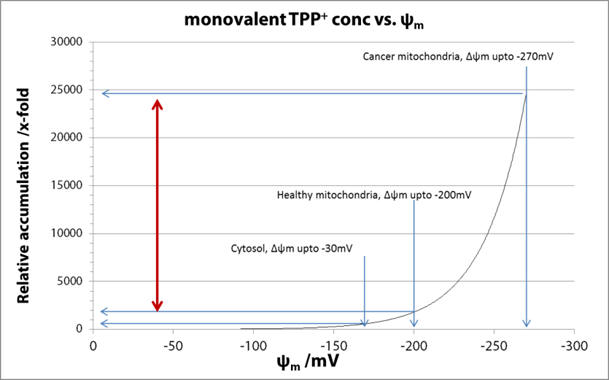
Graph 3 | Accumulation of monovalent cations to variously charged compartments
For monocations, the exponent in Equation 5 dictates a ca. ten-fold cation accumulation per 61.5mV difference. [39] Thus relative to the plasma the healthy mitochondria (-200mV) result in a several hundred-fold accumulate [40,41], and cancer mitochondria relative to plasma, at least an order of magnitude greater. [42,43] The selectivity depends upon the differences in membrane potentials and for a monocation the selectivity is 10-40x. See Figure 6 for a comparison of compartmental cation accumulation.
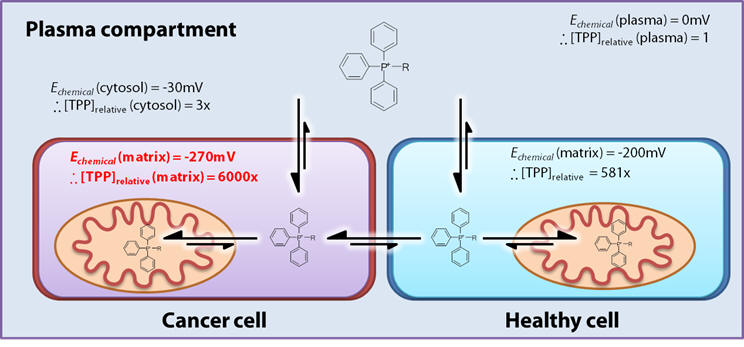
Figure 6 | Selective electrophoretic accumulation
A practical consequence of active accumulation is that exposure of cells to micromolar concentrations of TPP will result in millimolar matrix concentrations. TPPs rapidly partition into the mitochondria [45] such that cytosolic concentrations never rise to sufficiently high levels to become toxic.[64] This is extremely beneficial from the pharmacological perspective of limiting side-effects! The free matrix TPP concentration is likely to be 60-80% lower than the total mitochondrial concentration due to extensive interactions with the membrane. [46]
2.3 Uptake into the mitochondria – direct membrane traversal
Simple abundant physiological cations, certainly common metal cations cannot pass directly through the mitochondrial membrane. [47] Why are the movements of these ions rigorously controlled whereas TPP+s penetrate rapidly without mitochondrial control? To answer this question we must consider the cation solvation and factors that affect it.
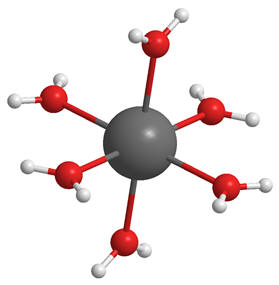
Figure 7 | First solvation sphere of a metal cation
Typical cations are strongly solvated and form solvation complexes (see Figure 7). Before membrane traversal can proceed total desolvation is required and this is highly endogonic involving loss of stabilising solvent interactions. The energy demand of desolvation can be estimated using the Born model which treats ions as point charges and the bulk solvent as a dielectric continuum. [48] The cations interact with the electric fields of the solvent molecules and these interactions constitute solvation. Thus the Born energy is a measure of solvation and is calculated using Equation 6. [49] The function has three variables, the charge (z), the solvent-adduct radius (r) and the dielectric (ε) of the solvent. For a discussion of electric permittivity see Appendix.
Equation 6 | The Born Equation
$$\Delta {G_{Born}} = \frac{1}{{8\pi {\varepsilon _0}}}\frac{{{z^2}}}{r}\left( {\frac{1}{{{\varepsilon _1}}} - \frac{1}{{{\varepsilon _2}}}} \right)$$
Born energy as a function of charge and radius (Graph 4) clearly illustrates the decreasing demand of desolvation with increasing radius and the enormously increasing demand with charge: large ions of low charge are most easily desolvated, whereas small ions, like metals are desolvated with difficulty. [50]
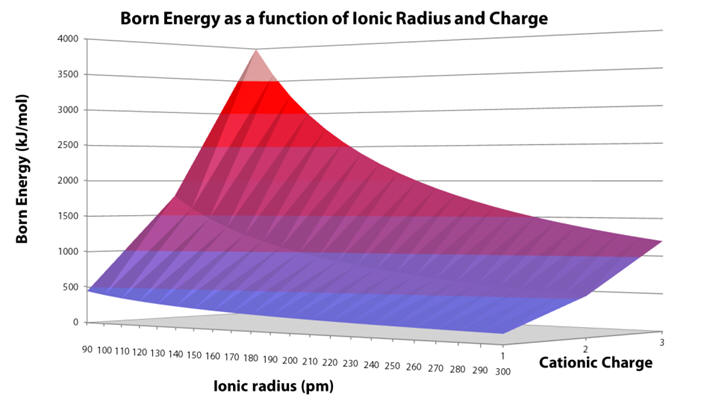
Graph 4 | Variation of solvation energy with ion charge and ionic radius
Triphenylphosphoniums are unlike typical physiological cations; the phenyl groups present a lipophilic accessibility surface. [51] They are unable to participate in H-bonding and they are not Lewis acidic. These factors result in poor phosphonium hydration and relative to metal ions TPPs are very easily desolvated. [52]
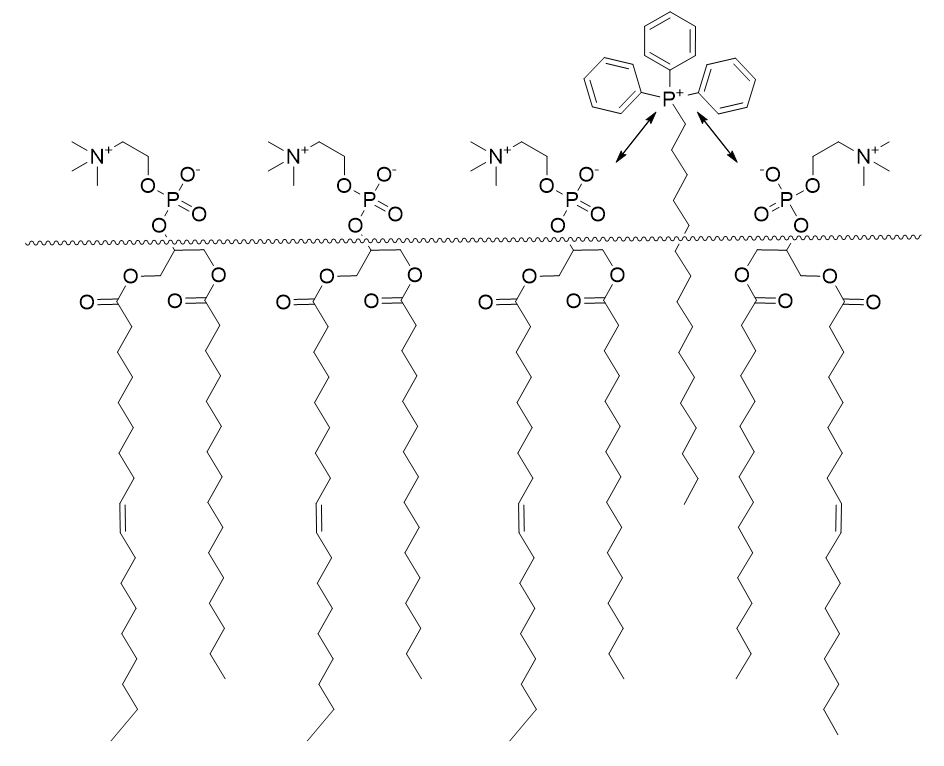
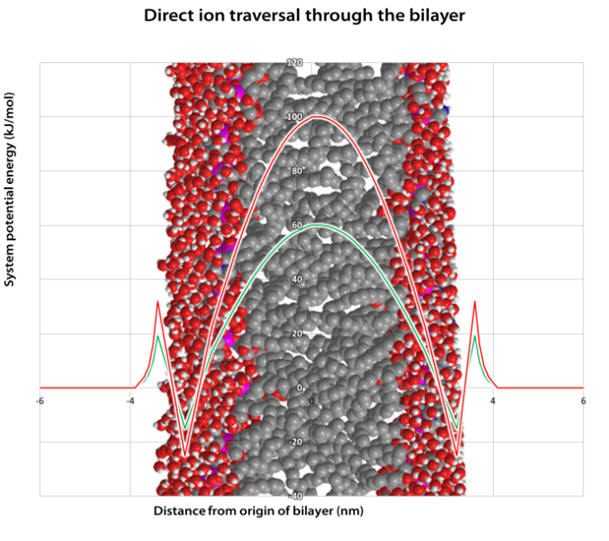
Once desolvated the ion is pulled through the membrane surface and into the core and as it goes, the Born energy varies (see Graph 5). The local maxima correspond to cation partial desolvation on approach to the membrane. The troughs correspond to stabilising interactions between the with the membrane lipid anionic phosphate (see Figure 9). This suggests there may be a propensity for monolayer adhesion of TPPs to the membrane surface which may contribute to the anti-cancer effect through membrane function modulation. The potential energy rapidly rises towards the membrane core due to increasingly unfavourable interactions due to a drop in permittivity, see Figure 8. [53]
 |
|
Graph 5 | Energy profile |
 |
|
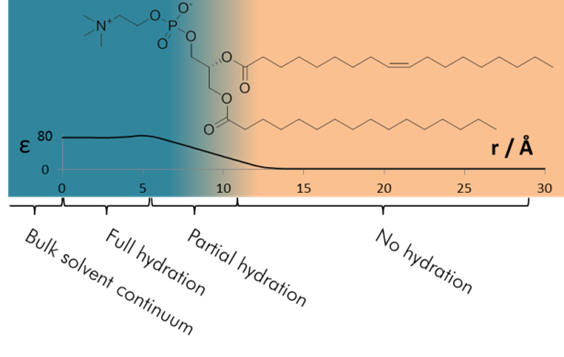
Figure 8 | Variation of permittivity [54] |
On the graph are shown the potential energies of two different cations, one with radius 116pm (red curve, Na+) and the other a radius of 300pm (green curve, an example organic cation). It is evident that the potential maxima is markedly lower for the larger cation which translates to a lower activation energy and a more facile membrane penetrating ability. [55] From this discussion it is clear that large ions of low charge density are more easily uptaken across membranes. Membrane penetration ability is therefore related to cation lipophilicity and larger ions more easily achieve it.
Figure 9 | Mitochondrial membrane embedding of DLCs