...on the rationalisation, design, optimisation and synthesis of novel phosphonium derived anti-cancer drugs
 |
Establishing Anti-Cancer Phosphonium Salt Structure-activity RelationshipsPart 4by Phil |
Design and Optimisation
4.1 Pre-clinical Candidate Optimisation
There are some very promising TPPs that look, with some optimisation to lend themselves to preclinical investigation.
Monocations
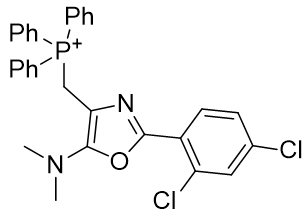
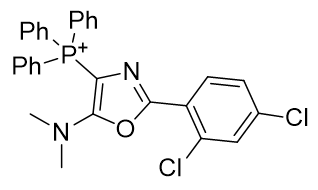
The lead candidate TP-197 (Table 5) has a methylene linker between the phosphonium and the substituent and there is room for optimisation up to the statistical CLogP limit of 8.5.
Table 5 | Lead candidate TP-197 (Neamati et. al.) and its desmethylene ‘parent’
 |
 |
|
| Compound | TP-197 | parent |
| CLogP | 7.65 | 7.43 |
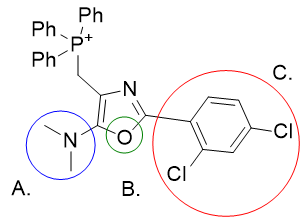
Analysis of the molecules reveals several sites for optimisation (indicated A, B and C).
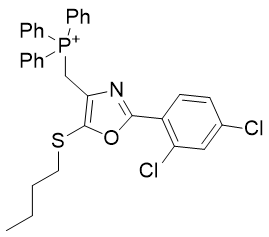
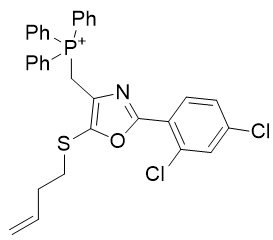
Table 6 | Optimisation candidates, TP-197-A and B
 |
 |
 |
|
| Derivative | TP-197-A | TP-197-B | |
| CLogP | 7.65 | 8.5 | 8.51 |
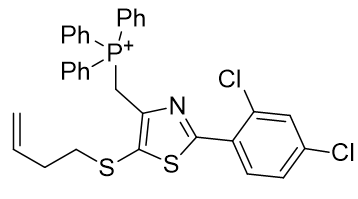
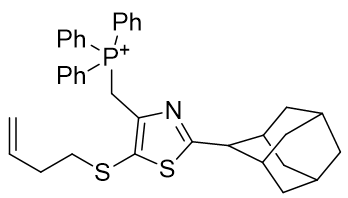
Substituent A is very hydrophilic. Substitution with butylthio affords the more lipophilic TP-197-A. De-saturation affords structure TP-197-B. Ring heteroatom B can be substituted with a heavier congener to afford the butylthio-thiazole heterocycle TP-197-C. Substituent C is already rather lipophilic, but the adamantyl group is more so and its incorporation affords the significantly more lipophilic candidate TP-197-D.
Table 7 | Optimisation candidates, TP-197-C and D
 |
 |
|
| Compound | TP-197-C | TP-197-D |
| CLogP | 8.7 | 9.52 |
Di-cations
The dications in literature are un-optimised structure but serve as proof of concept.
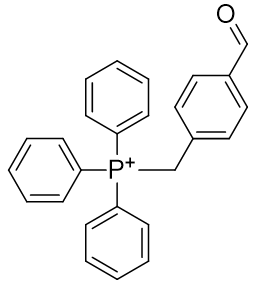
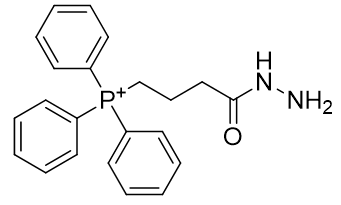
Table 8 | Lead candidate for dication phosphoniums
 |
 |
|
| Compound | TP-CHO | TP-HDZ |
| CLogP | 6.37 | 4.8 |
The LogP of both candidates can be improved, especially that of the hydrazide, TP-HDZ. Rapid in vivo assembly is desirable and so kinetic enhancements to the condensation mechanism can be pursued. It is preferable for the aldehyde to lack α-protons (potentially complicating reactivity by enabling competing routes), and so formylbenzene is a good structure to accept the hydrazide. It is important to refrain from introducing substituents that could potentially sterically impair the approach of nucleophilic hydrazide (Figure 24).
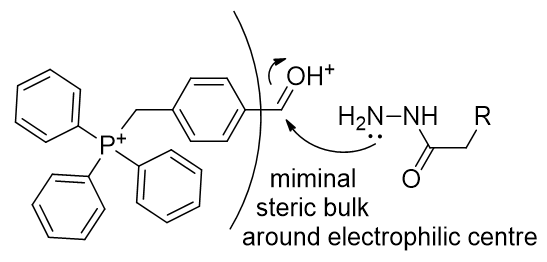
Figure 24 | Steric bulk around aldehyde best kept low
In course of the mechanism, δ- development at the carbonyl carbon and stabilisation of this would accelerate reactions. This can be achieved by introduction of electron withdrawing groups onto the benzene ring. The use of large chloro- groups carries the advantage of raising the logP to be within the ‘preferred range.’
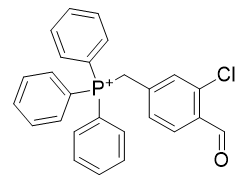
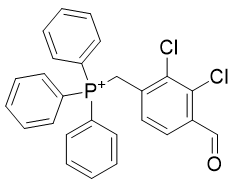
Table 9 | Optimisation candidate, TP-CHO-1 and TP-CHO-2
 |
 |
|
| Compound | TP-CHO-1 | TP-CHO-2 |
| CLogP | 7.02 | 7.38 |
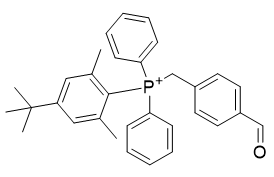
Whilst it may be anticipated that the 5,6-substitution pattern should not adversely affect the kinetics of reaction at carbonyl, effects cannot be ruled out. Instead substitution of the phenyl groups is attractive, an option entirely ignored by many literature reports. It is perhaps undesirable to alter the phenyl group as this obviates the use of cheap triphenylphosphine, necessitating the synthesis of custom phosphines. This avenue, if pursued opens access to more diverse means of optimisation. Replacement a phenyl ring with mesityl affords TP-CHO-3. Predictably the sym-t-butyl-m-xylene derivatives are more lipophilic.
Table 10 | Optimisation candidate, TP-CHO-3 and TP-CHO-4
 |
 |
|
| Compound | TP-CHO-3 | TP-CHO-4 |
| CLogP | 7.2 | 7.63 |
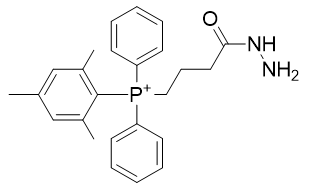
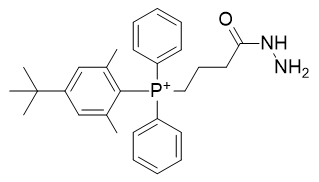
The same derivatisations are applicable to the hydrazide yielding TP-HDZ-1 and TP-HDZ-2; neither of these is particularly lipophilic. Fatty acids are highly lipophilic and commercially available and we can envisage a phosphonium derivative thereof. The docosahexaenoic acid derivative TP-HDZ-3 is extremely lipophilic.
Table 11 | Optimisation candidate, TP-HDZ-1, 2 and 3
 |
 |
|
| Compound | TP-HDZ-1 | TP-HDZ-2 |
| CLogP | 6.48 | 6.89 |
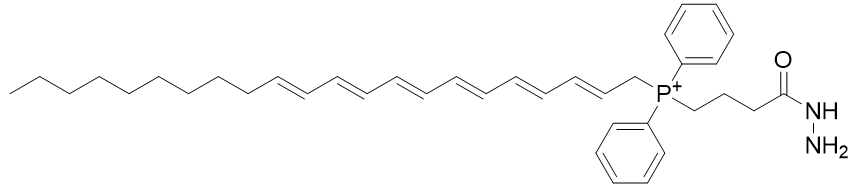 |
|
| Compound | TP-HDZ-3 |
| CLogP | 9.55 |
Alternative modes of self-assembly
Another coupling mechanism of interest is the Huisgen cycloaddition, a [2+3] addition reaction of azides to alkynes. [66]
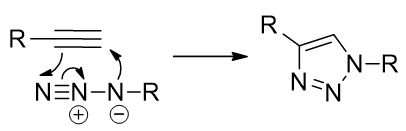
Scheme 2 | The Huisgen cycloaddition
These are an interesting choice because in the absence of good dipolariphiles, azides are invisible to many species [67] - their interaction requires specific orbital configurations (see Figure 25).
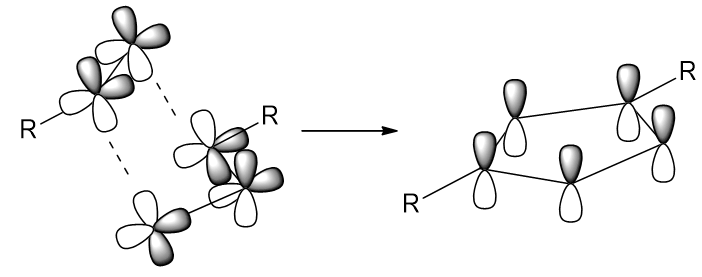
Figure 25 | Orbital interactions involved in the Huisgen cycloaddition
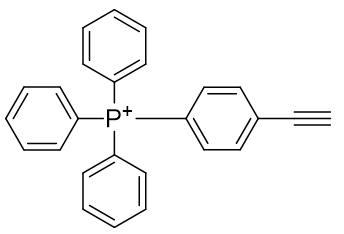
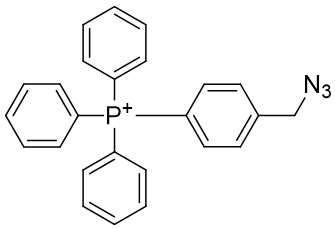
It would be interesting to see if this mode of attachment, one of the so called ‘Click’ reactions would facilitate effective assembly. In its basic form, this constitutes the new pair of agents, TP-HC2 and TP-HC3.
Table 12 | Experimental candidates for Huisgen click reaction
 |
 |
|
| Compound | TP-HC2 | TP-HC3 |
| CLogP | 6.65 | 5.39 |